Blowing up biology
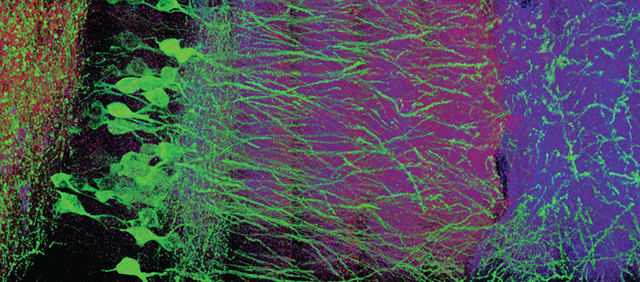
Part of a mouse brain viewed with expansion microscopy. The enlarged brain shows individual neurons, marked with green tags. (Courtesy of Edward Boyden.)
To solve a problem, sometimes you need to consider the exact opposite of what you think you know. Take trying to see the minute details of biological units, such as neurons in the brain. Instead of attempting to improve magnification on such a small scale, Massachusetts Institute of Technology engineer Edward Boyden asked, why not increase the size of the structure?
Boyden was wrestling with the limits of how much a structure can be magnified. Conventional microscopes rely on the physical properties of bending light. A beam of light can only be bent so far before the image gets fuzzy. To address this limitation, Boyden and colleagues use the power of expanding gels—the same kind of polymer used as an absorbent in diapers—to enlarge slices of preserved mouse brain and other nonliving biological structures.
Using Boyden’s methods, appropriately termed “expansion microscopy,” it is possible to enlarge structures up to five times their original size—with 20 times enlargement coming soon. Specific cells or structures in the preserved tissue are marked with different colored tags. And then, like in the investigation of a crime scene, the tissue is removed leaving a sort of chalk outline of colored tags. Because, the “biological copy machine” process removes all the original material, it can faithfully blow up the important landmarks without any stretches or tears.
Or as Boyden puts it, “we want to completely destroy everything so that nothing is destroyed at all.”
And now the outline, along with important biological connections, is large enough for researchers to see with conventional microscopes. The ability to use traditional microscopes is an advantage over other expensive high-magnification techniques. Boyden described his lab’s new methods to science writers Oct. 11 in the opening talk of CASW’s New Horizons in Science program at MIT during the ScienceWriters2015 conference.
Zooming in on disease mechanisms and therapies
And seeing how biological parts connect gives clues about how they work. To take a peek at brain structure, Boyden and colleagues tagged interesting points in slices of a mouse’s brain. Tagged brain cells can give insight to neurological disease. For instance, scientists can take advantage of the fact that different brain disorders produce signature proteins. Scientists can then locate cell types affected by the disorder from the protein tags. This ground truthing, Boyden explains, can help drive the development of new, more specific therapies.
Expansion microscopy has helped scientists explore more than just the brain. Because Boyden makes this tool freely available on the lab’s website, other researchers have used it to begin pinpointing molecules active in cancer cells, locating viruses within cells and identifying bacteria in the blood.
Another new frontier involves mapping the three-dimensional structure of DNA. It’s easy to think of DNA as a two-dimensional string. However, each meter-long copy of the genome curls up into a structure one-tenth the size of a human hair with unique twists and turns. Tightly curled DNA is less likely to be expressed than loose DNA, and it can be tricky to see the genetic structure.
“If you could look at the structure of the genome that would also tell you a lot about which genes are… regulated to be on or off,” Boyden said.
Imaging specific cells and molecules is just the beginning for the technology developed by Boyden’s lab. He considers future research that could map all the connections in a human brain and reconstruct it digitally.
“You could actually figure out what computations are happening when we see something, or when we make a decision, or when we have a feeling,” he says.
By combining expansion microscopy with other tools developed in the lab, Boyden has high hopes for future research. Using expansion microscopy, alongside methods that control the firing of individual neurons, scientists can begin pinpointing the nuts and bolts of how we work.
“The tools have gotten to the point now where I think we’re ready to start scalably mapping the mechanisms of life.”